November/December 2014
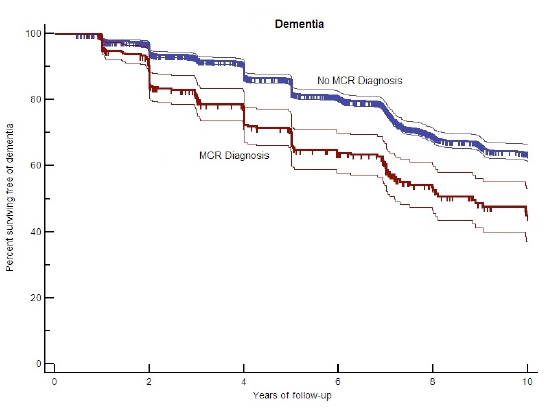
Gait Speed Reflects Cognitive Function As populations around the world age over the coming decades, the number of cases of dementia is projected to triple by 2050, creating a major public health concern across the globe.1 Identifying older adults early in the course of dementia offers the best opportunity to introduce preventive interventions to reduce the global dementia burden. Moreover, efficient case detection can also help identify modifiable risk factors for dementia. Gait and cognition are controlled by brain circuits that are vulnerable to multiple age-related pathologies such as vascular and neurodegenerative diseases. Hence, clinical strategies that help clinicians identify gait and cognitive deficits early can lead to improved risk prediction and improve management of high-risk older patients. Motoric Dysfunction Recently increasing evidence indicates that gait slowing occurs early in the course of dementia and may precede declines in cognitive tests, indicating that gait speed may be a sensitive marker of cognitive changes in aging.6 However, despite evidence of the link between cognitive and motor performance, there is a paucity of research capitalizing on these findings to improve dementia risk assessments. Cognitive Dysfunction and Predementia Syndromes Although many predementia syndromes have been previously described, most rely on procedures such as neuropsychological testing, biomarker assays, or neuroimaging. These procedures are costly and time consuming and may not be practical in many clinical settings around the world with limited resources. Furthermore, incidence and prevalence rates of various clinical predementia syndromes are inconsistent across the world. This is likely due to the broad guidelines of diagnosis that have not been adapted to account for social and cultural differences across countries.1 This is particularly apparent in low- and middle-income countries where a low prevalence of amnestic-MCI has been reported, and suggests that many high-risk seniors are not being identified by the current predementia syndromes.1 There exists a critical need to improve the accessibility, efficiency, and specificity of clinical dementia risk assessments for high-risk patients who can benefit from preventive measures. Motoric Cognitive Risk Syndrome Global Prevalence of MCR The association of MCR with major cognitive decline remained strong even when the analysis was restricted to a subset of very cognitively healthy participants with baseline MMSE scores greater than 27,9 supporting the concept that MCR was capturing very early stages of the dementia process. The figure below (derived from the recent global study9) illustrates that subjects with MCR at baseline were twice as likely to develop dementia over the next 10 years compared with those without an MCR diagnosis at baseline. In addition, subjects with MCR had more than twice the risk of developing Alzheimer's disease compared with those without MCR.
Adjustments to the individual criterion of MCR may help to refine, standardize, and enhance the current operational definition. For instance, instrumented gait assessments yield markers other than speed that could be used to improve the motoric criterion.10 However, utilization of instrumented methods comes at the cost of reduced accessibility in clinical settings but could be considered in research settings. In addition, we considered using a single uniform cutscore globally to define slow gait. Although this is a more simple strategy, single cutscores will not take into account variability in gait performance due to age, sex, or population differences.9 Self-report of mobility or walking difficulties suffers from lower sensitivity and specificity if being considered for use in defining slow gait.11 Cognitive complaints were assessed using a number of different standardized questionnaires in the global MCR prevalence study;9 however, use of a standardized method to assess subjective cognitive complaint questions, as well as use of informant reports may improve the specificity of this core MCR criterion. Clinical Use The MCR definition does not require cognitive tests for assigning the diagnosis. We have shown that there is partial overlap between participants diagnosed with MCR and MCI syndromes.8 These findings suggest that there are significant proportions of at-risk individuals who are not captured by any one predementia syndrome alone. Future studies could consider combining cognitive and motor criteria in settings where both these assessments are feasible to capture a larger pool of at-risk individuals. Treatment and Interventions What if there are no treatable diseases or causes for MCR? The diagnosis of MCR also allows clinicians time to recommend preventive strategies that might be effective in reducing the future risk of dementia. It also provides both patients and caregivers an opportunity to prepare with the knowledge that identified patients are at higher risk of developing dementia. For example, cognitive remediation approaches using computerized programs or cognitive training have demonstrated an improvement in higher level brain function in cognitively normal older adults. A small study of 24 frail older adults who participated in a computerized cognitive remediation program showed an improvement in gait velocity compared with their baseline performance, suggesting that cognitive remediation could be a new nonpharmacological way of improving mobility performance.14 Moreover, research has shown that low physical activity is associated with increased dementia risk,15 indicating the importance of keeping physically and mentally active to delay the onset of, or even prevent, cognitive decline and dementia. In general, the MCR concept provides a way of thinking about identifying risk and preventing dementia the same way we think about preventing other kinds of illnesses through maintaining a healthful lifestyle throughout the life course. — Emmeline Ayers, MPH, is the research coordinator for the Division of Cognitive and Motor Aging (Neurology) and Geriatrics (Medicine) at Albert Einstein College of Medicine in New York. — Joe Verghese, MBBS, is a professor of neurology and chief of the Integrated Divisions of Cognitive and Motor Aging (Neurology) and Geriatrics (Medicine) at Albert Einstein College of Medicine. References 2. Guralnik JM, Ferrucci L, Pieper CF, et al. Lower extremity function and subsequent disability: consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J Gerontol A Biol Sci Med Sci. 2000;55(4):M221-231. 3. Verghese J, Wang C, Lipton RB, Holtzer R, Xue X. Quantitative gait dysfunction and risk of cognitive decline and dementia. J Neurol Neurosurg Psychiatry. 2007;78(9):929-935. 4. Camicioli R, Howieson D, Oken B, Sexton G, Kaye J. Motor slowing precedes cognitive impairment in the oldest old. Neurology. 1998;50(5):1496-1498. 5. Holtzer R, Verghese J, Xue X, Lipton RB. Cognitive processes related to gait velocity: results from the Einstein Aging Study. Neuropsychology. 2006;20(2):215-223. 6. Buracchio T, Dodge HH, Howieson D, Wasserman D, Kaye J. The trajectory of gait speed preceding mild cognitive impairment. Arch Neurol. 2010;67(8):980-986. 7. Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004;256(3):183-194. 8. Verghese J, Wang C, Lipton RB, Holtzer R. Motoric cognitive risk syndrome and the risk of dementia. J Gerontol A Biol Sci Med Sci. 2013;68(4):412-418. 9. Verghese J, Annweiler C, Ayers E, et al. Motoric cognitive risk syndrome: multicountry prevalence and dementia risk. Neurology. 2014;83(8):718-726. 10. Hausdorff JM, Buchman AS. What links gait speed and MCI with dementia? A fresh look at the association between motor and cognitive function. J Gerontol A Biol Sci Med Sci. 2013;68(4):409-411. 11. Verghese J, Katz MJ, Derby CA, Kuslansky G, Hall CB, Lipton RB. Reliability and validity of a telephone-based mobility assessment questionnaire. Age Ageing. 2004;33(6):628-632. 12. Garde E, Mortensen EL, Krabbe K, Rostrup E, Larsson HBW. Relation between age-related decline in intelligence and cerebral white-matter hyperintensities in healthy octogenarians: a longitudinal study. Lancet. 2000;356(9230):628-634. 13. Rosano C, Brach J, Longstreth Jr WT, Newman AB. Quantitative measures of gait characteristics indicate prevalence of underlying subclinical structural brain abnormalities in high-functioning older adults. Neuroepidemiology. 2006;26(1):52-60. 14. Verghese J, Mahoney J, Ambrose AF, Wang C, Holtzer R. Effect of cognitive remediation on gait in sedentary seniors. J Gerontol A Biol Sci Med Sci. 2010;65(12):1338-1343. 15. Larson EB, Wang L, Bowen JD, et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann Intern Med. 2006;144(2):73-81. |