September/October 2018
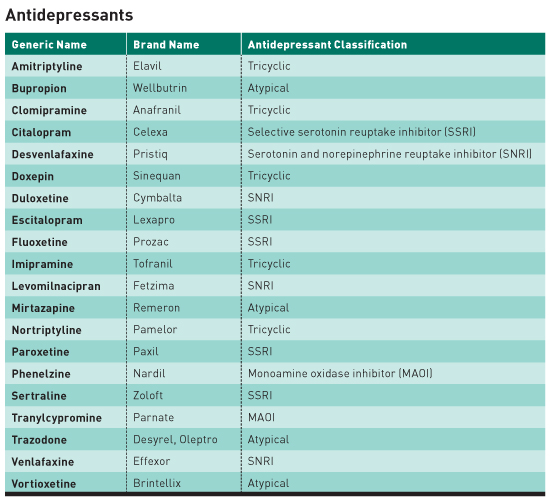
Medication Monitor: Challenges of Discontinuing Antidepressants Antidepressant discontinuation syndrome describes a cluster of symptoms that can occur following the discontinuation or lowering of an antidepressant dose.1 Typically these include flulike symptoms, insomnia, nausea, imbalance, sensory disturbances, and hyperarousal. These symptoms are generally mild and short-lived. However, for some patients, symptoms can be severe enough to make stopping antidepressants difficult.1,2 Health care professionals (HCPs) should understand the clinical implications of depression and antidepressant discontinuation syndrome. Furthermore, patients should be educated about the benefits and risks associated with antidepressant medications, including possible discontinuation symptoms. By taking preventive steps, HCPs can help reduce the occurrence and severity of discontinuation symptoms while improving the patient experience. Discontinuation Symptoms • Sensory symptoms: paresthesia or shocklike sensations sometimes described as "brain zaps" or "brain shivers." These also may include numbness and palinopsia (visual trails). Some describe a rushing noise in the head. • Disequilibrium or motor effect symptoms: dizziness, light-headedness, vertigo or feeling faint, restless legs, and gait instability. • General somatic symptoms: often compared with a flulike syndrome. This includes lethargy, headache, tremor, sweating, anorexia, and difficulty tolerating hot weather. • Affective symptoms: irritability, anxiety, low mood and tearfulness, possible suicidal thoughts, and visual hallucinations. • Gastrointestinal symptoms: nausea, vomiting, cramps, and diarrhea. • Sleep disturbances: insomnia, nightmares, and excessive dreaming. Onset of Symptoms and Severity For most patients, symptoms are mild and resolve without any intervention within one day to three weeks.2,3 If symptoms are severe, restarting the antidepressant or increasing back to the previous dose may be necessary, with symptoms usually resolving within 24 hours after reinitiating the antidepressant.2 Once symptoms have resolved, additional reduction attempts are possible, but with a slower and more cautious tapering approach.2 Antidepressant Pharmacology All classes of antidepressants (see table) can cause discontinuation symptoms with abrupt discontinuation.
• Selective serotonin reuptake inhibitors (SSRIs) primarily block the reuptake of serotonin. Common SSRIs include sertraline (Zoloft), escitalopram (Lexapro), and paroxetine (Paxil). • Serotonin and norepinephrine reuptake inhibitors (SNRIs) block the reuptake of serotonin and norepinephrine and include venlafaxine (Effexor) and duloxetine (Cymbalta). • Tricyclic antidepressants (TCAs) such as amitriptyline and nortriptyline primarily inhibit the reuptake of norepinephrine and, to a lesser degree, serotonin. TCAs also have anticholinergic properties. • Monoamine oxidase inhibitors (MAOIs) block the activity of monoamine oxidase, an enzyme involved in the breakdown of norepinephrine, serotonin, and dopamine. • Atypical antidepressants do not fall into one of these classes and have different mechanisms of action but affect norepinephrine, serotonin, and dopamine. An antidepressant's elimination half-life and metabolism are especially important. Discontinuation symptoms appear to be more prevalent and severe with antidepressants that have short half-lives.2 For example, the SSRI antidepressant paroxetine is more likely to cause discontinuation symptoms due to its short half-life (and less likely, if ever, with fluoxetine due to its long half-life).4 Of SNRIs, venlafaxine has a short-half life and is associated with a higher incidence of discontinuation symptoms.4 Discontinuation symptoms are similar among all classes of antidepressants; however, Parkinson's type symptoms and profound problems with balance appear to be especially prominent with tricyclic antidepressant discontinuation4 and are consistent with cholinergic rebound. Additionally, discontinuation symptoms with MAOIs may involve more serious symptomatology such as aggressiveness, agitation, catatonia, severe cognitive impairment, myoclonus, and psychotic symptoms and may require more intensive management.4,5 Weighing the Risks and Benefits The World Health Organization has identified major depression as the fourth-leading cause of disability worldwide and projects it will become the second-leading cause by 2020. Epidemiologic studies have shown that depression causes as much, and often more, physical disability and social and role impairment than most other chronic illnesses such as diabetes, arthritis, hypertension, and coronary artery disease. Major depression is also a common comorbidity in most other chronic conditions including heart disease, stroke, and diabetes mellitus.6 The link between depression and many chronic diseases is bidirectional. For example, preexisting depression is a predictor of future atherosclerotic coronary artery disease, cerebrovascular disease, diabetes, and osteoporosis, and, conversely, having a significant physical illness predicts higher prevalence of major depression compared with individuals without the physical illness. Depression can lead to worse outcomes in patients with heart disease (eg, coronary artery disease, congestive heart failure), including increased risk of reinfarction following MI and up to a threefold increase in all-cause mortality. Another example is that patients with diabetes and depression have worse glycemic control, more microvascular and macrovascular complications, and greater all-cause mortality.6 Education The FINISH mnemonic (Flulike Symptoms, Insomnia, Nausea, Imbalance, Sensory Disturbances, Hyperarousal) is a useful tool to describe antidepressant discontinuation symptoms to patients.2 Prescribers should be able to recognize the differences between possible relapse of a patient's originally treated condition and discontinuation symptoms to avoid continuing an antidepressant unnecessarily. Whereas symptoms indicating depression relapse typically occur two to three weeks after stopping an antidepressant, discontinuation symptoms can occur in as little as a few hours after missing a dose.2 Educate patients about the importance of being adherent to their treatment regimens, as missing even one or two doses of some short-acting antidepressants could result in withdrawal symptoms, leading them to believe their treatment is ineffective. This could also result in unnecessary changes in their antidepressant therapy.2 Switching antidepressants may also result in discontinuation syndrome from stopping the first agent, leading patients to associate these symptoms as adverse effects from the new antidepressant, resulting in more therapy changes.2 Tapering While there are conflicting opinions about the optimal taper schedule or duration, most recommendations call for tapering down over at least four weeks. Restarting the antidepressant or increasing the dose may be appropriate if symptoms are severe.2 For patients taking antidepressants with shorter half-lives, such as venlafaxine and paroxetine, a more gradual dose reduction is advisable due to the greater potential for severe discontinuation syndrome.9 — Mark D. Coggins, PharmD, BCGP, FASCP, is vice president pharmacy services and medication management for skilled nursing centers operated by Diversicare in 10 states and is a past director on the board of the American Society of Consultant Pharmacists. He was nationally recognized by the Commission for Certification in Geriatric Pharmacy with the 2010 Excellence in Geriatric Pharmacy Practice Award. References 2. Haddad P, Anderson I. Recognizing and managing antidepressant discontinuation symptoms. Adv Psychiatr Treat. 2007;13(6):447-457. 3. Schweitzer I, Maguire K. Stopping antidepressants. Aust Prescr. 2001;24(1):13-15. 4. Garner EM, Kelly MW, Thompson DF. Tricyclic antidepressant withdrawal syndrome. Ann Pharmacother. 1993;27(9):1068-1072. 5. Curtin F, Berney P, Kaufmann C. Moclobemide discontinuation syndrome predominantly presenting with influenza-like symptoms. J Psychopharmacol. 2002;16(3):271-272. 6. Cohen A; World Health Organization, Regional Office for Europe. Addressing comorbidity between mental disorders and major noncommunicable diseases: background technical report to support implementation of the WHO European Mental Health Action Plan 2013–2020 and the WHO European Action Plan for the Prevention and Control of Noncommunicable Diseases 2016–2025. http://www.euro.who.int/__data/assets/pdf_file/0009/342297/Comorbidity-report_E-web.pdf. Published 2017. 7. van Geffen EC, Hugtenburg JG, Heerdink ER, van Hulten RP, Egberts AC. Discontinuation symptoms in users of selective serotonin reuptake inhibitors in clinical practice: tapering versus abrupt discontinuation. Eur J Clin Pharmacol. 2005;61(4):303-307. 8. Zarowitz BJ. Antidepressant tapering: avoiding adverse consequences of gradual dose reduction. Geriatr Nurs. 2007;28(2):75-79. 9. Tint A, Haddad PM, Anderson IM. The effect of rate of antidepressant tapering on the incidence of discontinuation symptoms: a randomised study. J Psychopharmacol. 2008;22(3):330-332. |