September/October 2019
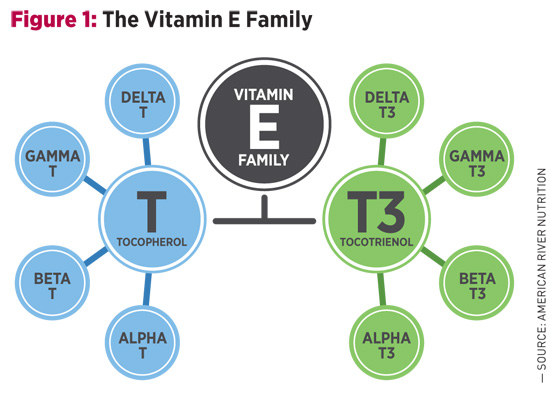
There’s More Than One Type of Vitamin E — The Study of Tocotrienol and Chronic Conditions Known as an essential nutrient, a vitamin is a compound the body does not make but that is required in minute amounts to support growth and development. If the vitamin is not obtained from outside sources, serious deficiencies and illnesses can result. Most vitamin deficiencies have a clear correlation with certain disorders. It’s known, for example, that lack of vitamin C causes scurvy, as first documented by Hippocrates, while the symptoms of rickets were reported as early as the first few centuries CE and were later linked to vitamin D deficiency. But why do individuals take vitamin E? To explore why vitamin E became an essential nutrient, it’s worthwhile to take a step back in history to its original discovery in 1922.1 At the time, Herbert McLean Evans and Katherine Bishop, anatomists from the University of California, Berkeley, were experimenting with different diet regimens in rats. They noted that animals did extremely well on a standard diet of fat, carbohydrates, protein, salt, and vitamins A and B, but despite their healthy appearance were completely sterile. Ascribing this deficit to dietary shortcomings, the researchers added green leafy vegetables to the animals’ diet, inadvertently reintroducing the missing element known today as vitamin E. Evans isolated the vitamin from wheat germ oil in 1936, and proposed naming the compound alpha-tocopherol, stemming from the Greek words tokos (“childbirth”) and phero (“to bear”), with the “-ol” indicating an alcohol group that is also the site of its antioxidant activity.2 Soon, it became clear that alpha-tocopherol was not the only form of vitamin E, although it’s still the form people most often associate with the term. As seen in Figure 1, other forms of vitamin E include alpha-, beta-, gamma-, and delta-tocopherol, and alpha-, beta-, gamma-, and delta-tocotrienol. These compounds are similar in molecular makeup, but slight nuances allow for a variety of functional differences among the group.
One characteristic that unites them—by way of their alcohol group—is their ability to offer protection from oxidation by donating electrons to free radicals, particularly in membranes and lipoproteins (eg, LDL cholesterol). With more than 38 trillion cells (all surrounded by cell membranes) in the human body, this is no small feat. In fact, more than 90% of cell membrane antioxidants are in the form of vitamin E.3 While much of the focus has been on the alpha-tocopherol form of vitamin E, it’s now known that tocotrienol—because of its unique molecular makeup—is approximately 50-fold more active as an antioxidant.4 Tocotrienols are included in clinical trials ranging from those for cardiovascular disease to cancer, with one study showing a doubling of life expectancy in women with advanced-stage ovarian cancer.5 Although research on tocotrienols is well underway, few have heard of this important, albeit elusive, member of the vitamin E family. Tocotrienols were first discovered in the late 1950s and—originally thought to be tocopherols—were mislabeled with Greek letters such as epsilon and zeta.6 The mistake was recognized,7,8 but official designations in the Merck Index were not corrected until 2001.9,10 It’s not surprising, then, that tocotrienols are only now emerging into public view, and the timing is fortuitous. According to the US Census Bureau, older adults (age 65 and older) will soon outnumber children, and with increasing age comes a heightened risk of chronic conditions. Tocotrienols may help prevent and address aging concerns through their antioxidant properties. The breadth of the vitamin E family is such that it cannot simply be reduced to the task of an essential nutrient. First discovered as the birth vitamin, its benefits extend well into potent antioxidant applications. Emerging research points to tocotrienol’s role as an antiaging nutrient helpful for addressing chronic conditions. Shape Matters
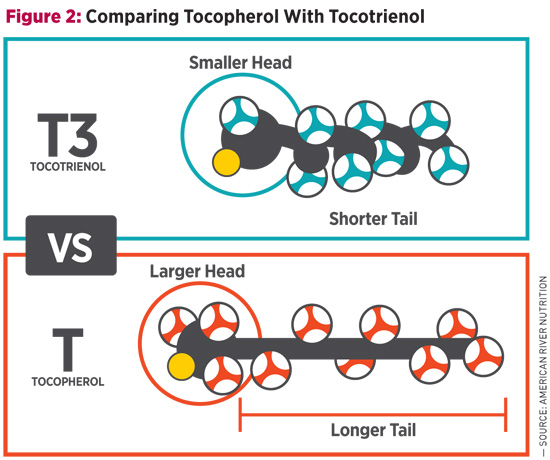
Between the four members (alpha-, beta-, gamma-, delta-) of both tocopherols and tocotrienols, there are shape differences as well, and these are found in the head region of the molecule. Alpha- forms have the largest heads, whereas delta- forms have the smallest; beta- and gamma- are somewhere in between. Since it’s more difficult for larger molecules to gain access to the cell membrane, smaller molecules are thought to be more active. This makes delta-tocotrienol—the smallest of the eight vitamin E molecules11,12—the most effective cell membrane antioxidant and is likely the reason for its therapeutic potential found in clinical trials. Vitamin E’s Flaw Alpha-tocopherol, which is the most prominent vitamin E in dietary supplements such as multivitamins, not only reduces blood levels of other tocopherols and tocotrienols,15-17 but also interferes with their functional benefits,18,19 including tocotrienol’s ability to regulate lipid levels.20-22 Moreover, when taken in high doses, alpha-tocopherol seems to do the opposite of what many think it promises. Numerous studies on alpha-tocopherol have shown that it can increase the risk of certain cancers,23,24 raise blood pressure,25,26 and elevate lipids.27,28 Consider the Selenium and Vitamin E Cancer Prevention Trial (SELECT), for example. Men were expected to have a reduced risk of prostate cancer with supplementation, but instead their risk increased by 17%.23,24 Of further note is a recently published evaluation of a subset of the Women’s Health Initiative, which showed that alpha-tocopherol supplementation was associated with an increased risk of cardiovascular disease.29 Vitamin E Sources The majority of supplemental tocopherols are derived from soy, with the United States being the top producer of the crop. Soy is naturally high in gamma-tocopherol, so when alpha-tocopherol is required, as is typical for most multivitamins, manufacturers convert soy-derived gamma-tocopherol to alpha-tocopherol. This means that most alpha-tocopherol supplements are not natural, but at best semisynthetic. With the main advantage of synthetic vitamin E being low cost, its use clinically is less desirable, partially because these forms are alien to the human body. Large trials conducted using synthetic versions of alpha-tocopherol, including SELECT and the Physician’s Health Study II, among others, yielded unfavorable results.23,30,31 With sufficient levels of alpha-tocopherol available through the diet and occurrences of vitamin E deficiencies being rare, supplementation may be less crucial than originally thought. Whereas readily available dietary sources such as seeds, nuts, oils, vegetables, and fortified foods supply plenty of tocopherols, this is not the case for tocotrienols. On the supplemental side, palm and rice bran oils were first identified as major sources of tocotrienol, but they carry the drawback of containing significant amounts of alpha-tocopherol, which interferes with tocotrienol functions and benefits. Tocotrienol availability improved in 2001, when annatto, an Amazonian rainforest plant, was found to contain only tocotrienols. Annatto Tocotrienol Clinical studies on annatto tocotrienol cover a range of applications relevant to antiaging medicine, from cardiometabolic health to bone strengthening and cellular well-being. The following is an overview with guidance on doses. Cardiovascular Benefits Not only do tocotrienols reduce cholesterol, but they have also been shown to lower triglyceride levels and high-sensitivity C-reactive protein (hs-CRP).33 Since alpha-tocopherol repeatedly displayed an inhibitory effect on tocotrienol lipid-lowering benefits, it’s recommended to supplement a formula that is tocopherol-free, such as from annatto, at an approximate dose of 200 mg/day.34 Liver Health Bone Strengthening The study, designed to test annatto tocotrienol’s bone-promoting benefits in postmenopausal women with osteopenia, showed that 300 mg of the vitamin significantly improved bone turnover after three months.37 More specifically, biomarkers for bone building increased by as much as 115%, whereas bone resorption (or breakdown) markers decreased as much as 24%. Annatto tocotrienol’s antioxidant capacity is thought to be one major mechanism at work, with oxidative stress decreasing up to 49%. Cellular Well-Being In another phase 2 clinical trial in women with advanced-stage ovarian cancer, 300 mg annatto tocotrienol three times per day—given in combination with chemotherapy drug Avastin—significantly improved life expectancy.5 Whereas Avastin typically results in patient survival without disease progression of two to four months and overall survival of five to seven months, the combination with annatto tocotrienol allowed for progression-free and overall survival of 6.9 months and 10.9 months, respectively, essentially doubling the life expectancy. Annatto tocotrienols are currently used in additional phase II clinical trials on cancers of the ovary, breast, colon, and lung, conducted at Vejle Hospital in Denmark. Most of these studies are slated to be completed within the next year or two. For supplementation purposes, it’s important to remember that annatto tocotrienol is fat soluble and should always be taken with a meal. Due to known interference issues, it’s also recommended to take tocotrienols at least six hours apart from alpha-tocopherol. Successful in clinical trials and with functions that go beyond its antioxidant properties, annatto tocotrienol conveys a broad range of antiaging benefits that may help address a number of today’s demanding health concerns. — Anne Trias, MS, received her master of science in microbiology from the University of Massachusetts, Amherst. For the past 15 years, her work has focused on the science and development of tocotrienol as a rising ingredient in the industry, and she’s written numerous articles and editorial contributions on the subject of vitamin E. Anne Trias, MS, reports the following relevant disclosure: She is product director at American River Nutrition (americanrivernutrition.com), producer of DeltaGold® tocotrienols sourced from the annatto plant.
References 2. Evans HM, Emerson OH, Emerson GA. The isolation from wheat germ oil of an alcohol, alpha-tocopherol, having the properties of vitamin E. J Bio Chem. 1936;113:319-332. 3. Esterbauer H, Puhl H, Waeg G, Krebs A, Dieber-Rotheneder M. The role of vitamin E in lipoprotein oxidation. In: Packer L, Fuchs J, eds. Vitamin E in Health and Disease. New York, NY: Marcel Dekker; 1993:649-671. 4. Serbinova E, Kagan V, Han D, Packer L. Free radical recycling and intramembrane mobility in the antioxidant properties of alpha-tocopherol and alpha-tocotrienol. Free Radic Biol Med. 1991;10(5):263-275. 5. Thomsen CB, Andersen RF, Steffensen KD, Adimi P, Jakobsen A. Delta tocotrienol in recurrent ovarian cancer. A phase II trial. Pharmacol Res. 2019;141:392-396. 6. Bunyan J, Green J, Mamalis P, Marcinkiewicz S. Biological potency of zeta-tocopherol. Nature. 1957;179(4556):418-419. 7. Bunyan J, McHale D, Green J, Marcinkiewicz S. Biological potencies of epsilon- and zeta-1-tocopherol and 5-methyltocol. Br J Nutr. 1961;15:253-257. 8. Pennock JF, Hemming FW, Kerr JD. A reassessment of tocopherol in chemistry. Biochem Biophys Res Commun. 1964;17(5):542-548. 9. Tocols: 9631 through 9638. In: Merck Index. 12th ed. Rahway, NJ: Merck Publishing Group; 1996:1620-1621. 10. Tocols: 9570 through 9577. In: Merck Index. 13th ed. Rahway, NJ: Merck Publishing Group; 2001:1693-1694. 11. Atkinson J, Epand RF, Epand RM. Tocopherols and tocotrienols in membranes: a critical review. Free Radic Biol Med. 2008;44(5):739-764. 12. Muller L, Theile K, Böhm V. In vitro antioxidant activity of tocopherols and tocotrienols and comparison of vitamin E concentration and lipophilic antioxidant capacity in human plasma. Mol Nutr Food Res. 2010;54(5):731-742. 13. Trias AM, Tan B. Alpha-tocopherol: a detriment to tocotrienol benefits. In: Tan B, Watson RR, Preedy VR, eds. Tocotrienols: Vitamin E Beyond Tocopherols. 2nd ed. Boca Raton, FL: CRC Press; 2013:61-78. 14. Kostic D, White WS, Olson JA. Intestinal absorption, serum clearance, and interactions between lutein and beta-carotene when administered to human adults in separate or combined oral doses. Am J Clin Nutr. 1995;62(3):604-610. 15. Drotleff AM, Bohnsack C, Schneider I, Hahn A, Ternes W. Human oral bioavailability and pharmacokinetics of tocotrienols from tocotrienol-rich (tocopherol-low) barley oil and palm oil formulations. J Funct Foods. 2014;7:150-160. 16. Handelman GJ, Machlin LJ, Fitch K, Weiter JJ, Dratz EA. Oral alpha-tocopherol supplements decrease plasma gamma-tocopherol levels in humans. J Nutr. 1985;115(6):807-813. 17. Huang HY, Appel LJ. Supplementation of diets with alpha-tocopherol reduces serum concentrations of gamma- and delta-tocopherol in humans. J Nutr. 2003;133(10):3137-3140. 18. Guthrie N, Gapor A, Chambers AF, Carroll KK. Inhibition of proliferation of estrogen receptor–negative MDA-MB-435 and –positive MCF-7 human breast cancer cells by palm oil tocotrienols and tamoxifen, alone and in combination. J Nutr. 1997;127(3):544S-548S. 19. Peralta EA, Brewer AT, Louis S, Dunnington GL. Vitamin E increases biomarkers of estrogen stimulation when taken with tamoxifen. J Surg Res. 2009;153(1):143-147. 20. Khor HT, Ng TT. Effects of administration of alpha-tocopherol and tocotrienols on serum lipids and liver HMG CoA reductase activity. Int J Food Sci Nutr. 2000;51 Suppl:S3-S11. 21. Qureshi AA, Pearce BC, Nor RM, Gapor A, Peterson DM, Elson CE. Dietary alpha-tocopherol attenuates the impact of gamma-tocotrienol on hepatic 3-hydroxy-3-methylglutaryl coenzyme A reductase activity in chickens. J Nutr. 1996;126(2):389-394. 22. Shibata A, Kawakami Y, Kimura T, Miyazawa T, Nakagawa K. α-tocopherol attenuates the triglyceride- and cholesterol-lowering effects of rice bran tocotrienol in rats fed a Western diet. J Agric Food Chem. 2016;64(26):5361-5366. 23. Klein EA, Thompson IM Jr, Tangen CM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306(14):1549-1556. 24. Lippman SM, Klein EA, Goodman PJ, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2009;301(1):39-51. 25. Li Z, Evans C, Cade J. Dietary vitamin E intake and blood pressure in UK adolescents: a longitudinal study. Paper presented at: American Society for Nutrition Annual Meeting; 2018; Boston, MA. 26. Miyamoto K, Shiozaki M, Shibata M, Koike M, Uchiyama Y, Gotow T. Very-high-dose alpha-tocopherol supplementation increases blood pressure and causes possible adverse central nervous system effects in stroke-prone spontaneously hypertensive rats. J Neurosci Res. 2009;87(2):556-566. 27. Khor HT, Chieng DY, Ong KK. Tocotrienols inhibit liver HMG CoA reductase activity in the guinea pig. Nutr Res. 1995;15(4):537-544. 28. Howard DR, Rundell CA. Vitamin E and serum lipids: a non-correlation. Am J Clin Pathol. 1982;77:243-244. 29. Prentice RL, Pettinger M, Neuhouser ML, et al. Application of blood concentration biomarkers in nutritional epidemiology: example of carotenoid and tocopherol intake in relation to chronic disease risk. Am J Clin Nutr. 2019;109(4):1189-1196. 30. Sesso HD, Buring JE, Christen WG, et al. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2008;300(18):2123-2133. 31. Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142(1):37-46. 32. Pearce BC, Parker RA, Deason ME, Qureshi AA, Wright JJ. Hypocholesterolemic activity of synthetic and natural tocotrienols. J Med Chem. 1992;35(20):3595-3606. 33. Pervez MA, Khan DA, Ijaz A, Khan S. Effects of delta-tocotrienol supplementation on liver enzymes, inflammation, oxidative stress and hepatic steatosis in patients with nonalcoholic fatty liver disease. Turk J Gastroenterol. 2018;29(2):170-176. 34. Houston M. The role of nutraceutical supplements in the treatment of dyslipidemia. J Clin Hypertens (Greenwich). 2012;14(2):121-132. 35. Nguyen P, Valanejad L, Cast A, et al. Elimination of age-associated hepatic steatosis and correction of aging phenotype by inhibition of cdk4-C/EBPα-p300 axis. Cell Rep. 2018;24(6):1597-1609. 36. Khallouki F, de Medina P, Caze-Subra S, et al. Molecular and biochemical analysis of the estrogenic and proliferative properties of vitamin E compounds. Front Oncol. 2016;5:287. 37. Shen CL, Yang S, Tomison MD, Romero AW, Felton CK, Mo H. Tocotrienol supplementation suppressed bone resorption and oxidative stress in postmenopausal osteopenic women: a 12-week randomized double-blinded placebo-controlled trial. Osteoporos Int. 2018;29(4):881-891. 38. Cinar D, Tas D. Cancer in the elderly. North Clin Istanb. 2015;2(1):73-80. 39. Springett GM, Husain K, Neuger A, et al. A phase I safety, pharmacokinetic, and pharmacodynamic presurgical trial of vitamin E δ-tocotrienol in patients with pancreatic ductal neoplasia. EBioMedicine. 2015;2(12):1987-1995. |